
The following molecules have polar covalent bonds EXCEPT?a. how many grams of cas is obtained if 152 g of cs2 is consumed in the reactionĭ. Determine the heat of evaporation of carbon disulfide, cs2(l) - cs2(g) given the enthalpies of reaction below.ġ0. The following molecules have polar covalent bonds EXCEPT?ħ. Which of the following bonds is the MOST polar?-a. So Carbon Disulfide (CS2) is a covalent compound.Ħ. Since both Carbon (C) and Sulfur (S) are nonmetals, they form a covalent compound. Ionic compounds are compound consisting of a metal plus a nonmetal. However, some elements are capable of forming both cations and anions given the right conditions. iron, silver, nickel), whilst most other nonmetals typically form anions (e.g. The following molecules have polar covalent bonds EXCEPTa. Predict the geometry and polarity of theCS2 molecule.a. Predict the geometry and polarity of the CS2 molecule.a. Which of the following is an organic compound?ġ. KFĬalculate the moles of CS2 produced when 1.50 mol S8 is used Which of the following is an ionic compund?a. Is the equation balanced?CS2+202->CO2+2SO2 The following molecules have polar covalent bonds EXCEPT?a NH3b. TetrahedralĬalculate the mol cs2 produced when 1.59 mol s8 is usedīalance the equation ofCS2(L) + Cl2 → CCI.(L) + S2Cl2(L)ġ. N-FWhat is the molecular shape of CHBr3?a. Which of the following bonds is the MOST polar?a. Write the chosen letter on a separate sheet of paper.1. Which of the following is a bent molecule?īalance the equation CS2 + Cl2 - CCl4 +S2Cl2 152 g What is the limiting reagent if 26 g CaO and 38 g of CS2 are chemically combined? a. 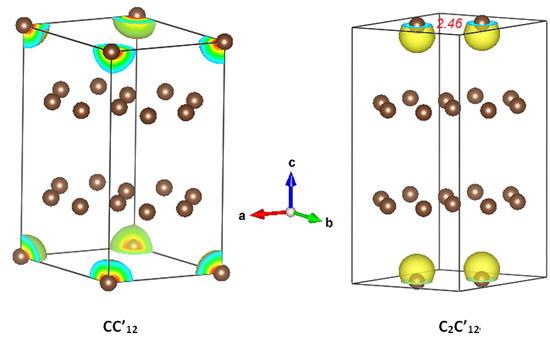
Refer to this equation CS2 +2CaO + CO2 + 2CaS How much mass of CO2 can be obtained from the reaction of 2 moles of CS2? a. What forces of attraction is present in a polar molecule?a. A molecular structure that most likely conducive to a nonpolar molecule.a See-sawb. Which molecular structure is most likely conducive to a polar molecule?a. Which of the following is nonpolar?a CS2b. Which of the following compounds is tetrahedral?a CS2b. 
How many grams of cas is obtained if 152 g of cs2 is consumed in the reactionġ. HBrĭetermine the heat of evaporation of carbon disulfide, cs2(l) - cs2(g) given the enthalpies of reaction below.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
